Traitwell Releases Gene-Drug Interaction App: The Promise of Pharmacogenomics
It's time to put information on drug-gene interactions into everyone's hands.
You assume that meds will just work—until the specific medication you need doesn’t work for you. Then it might take weeks or months of miserable trial and error and unpleasant side effects before you chance upon the right drug to take care of your original health issue – and that’s with your doctor’s help!
Pharmacogenomics—an area of precision medicine—is a promising development that may help each of us understand our unique responses to specific medications so that we can get to the right medication faster. That’s because your genome plays an important role in how your body processes and responds to medications, along with your age, weight, health conditions, and other medications. Pharmacogenomics focuses on the genetic variants that may affect how people metabolize and respond to different drugs. That means that as we learn more about drug-gene interactions, experiencing side effects might one day be a thing of the past.

There are significant resources already out there for understanding drug-gene interactions. Both patients and healthcare providers are increasingly interested in and have more access to pharmacogenomic testing, through the increasing availability of direct-to-consumer pharmacogenetic testing and the broader use of pharmacogenetic panels. Gene-based drug prescription recommendations are easily available online thanks to Food and Drug Administration-approved drug labeling and Clinical Pharmacogenetics Implementation Consortium guidelines.
Previous surveys found that people are very interested in pharmacogenomic testing for all the reasons you’d expect: prediction of (serious) side effects, guidance for drug dosage, and a better drug selection process, all customized to the needs of the individual. You don’t need to be a medical professional to understand that drug treatments that work for most people may not work for you, and if your own genes can provide valuable information for tailoring drug treatments to your unique needs, then pharmacogenomics is very interesting, very valuable, and very necessary.
Accordingly, most people want pharmacogenomics results in the hands of their healthcare providers. A 2016 survey found that 92% of respondents would share their results with their healthcare providers. A 2020 survey found that 74% planned to discuss their results with their healthcare providers and that 37% actually did so at a follow-up appointment. To meet patient demand and take advantage of the increasing utility of pharmacogenomic research, more and more healthcare systems are implementing pharmacogenomics programs, and statewide initiatives to implement PGx testing are increasingly underway.
Pharmacogenomic testing will one day become part of the medical standard of care.
In May of 2022, Oregon Health and Science University agreed to pay one million dollars to settle a lawsuit. The lawsuit asserted that the university's negligence had killed a cancer patient. The negligence involved the failure to test for a known genetic condition. This genetic condition prevented the cancer patient from correctly processing the chemotherapy drug he received from the hospital. The genetic condition affects as many as 8% of people, and it is possible to counteract the adverse reaction to the drug if an antidote is given in time. Because the hospital did not tell the patient or his now-widow about the genetic condition, did not test for the genetic condition, and therefore did not act quickly enough to his adverse drug-gene interaction, the patient died.
As part of the settlement, the university will now be required to tell patients about this genetic condition before initiating this chemotherapy drug.
Medical treatments that hurt instead of help people are a known problem. Harmful or suboptimal medications, vaccines, and related therapies are associated with avoidable illnesses and mortality affecting millions of Americans, accounting for approximately 2 million hospital stays, 3.5 million physician office visits, 1 million emergency department visits, and 125,000 hospital admissions each year. These “nonoptimized medication therapies” cost the U.S. health sector from $495.3 billion to $672.7 billion in potentially avoidable spending in 2016, with direct costs of the medications costing an additional $308 billion in 2020.
Pharmacogenomics is about identifying your genetic attributes related to adverse or ineffective drug responses. As the cost of genetic testing continues to drop and public knowledge about genomics continues to rise, pharmacogenomics will be a major part of confronting the largest population health challenges in the U.S. healthcare sector.
Alas, you can't get pharmacogenomics testing without a doctor. Your doctor would need to order such a test and explain drug-gene interactions to you—assuming, of course, that your doctor knows about pharmacogenomic testing and thinks it’s necessary for your case.
To meet the need for consumer-friendly pharmacogenomics, we’re launching Traitwell PGX.
Despite widespread availability of direct-to-consumer DNA tests like 23andMe and Ancestry DNA, most people think these tests are only useful for genealogy and ancestry, not for informing healthcare concerns. That’s due to a lack of good direct-to-consumer pharmacogenomics reports.
We built Traitwell PGX to empower average Americans with their own pharmacogenomic test results. Not too long ago, average Americans demanded more forensic DNA evidence—due in no small part to shows like CSI—and forced the criminal justice system to accept technological progress. Traitwell PGX will inspire average Americans to do the same for pharmacogenomics in healthcare.
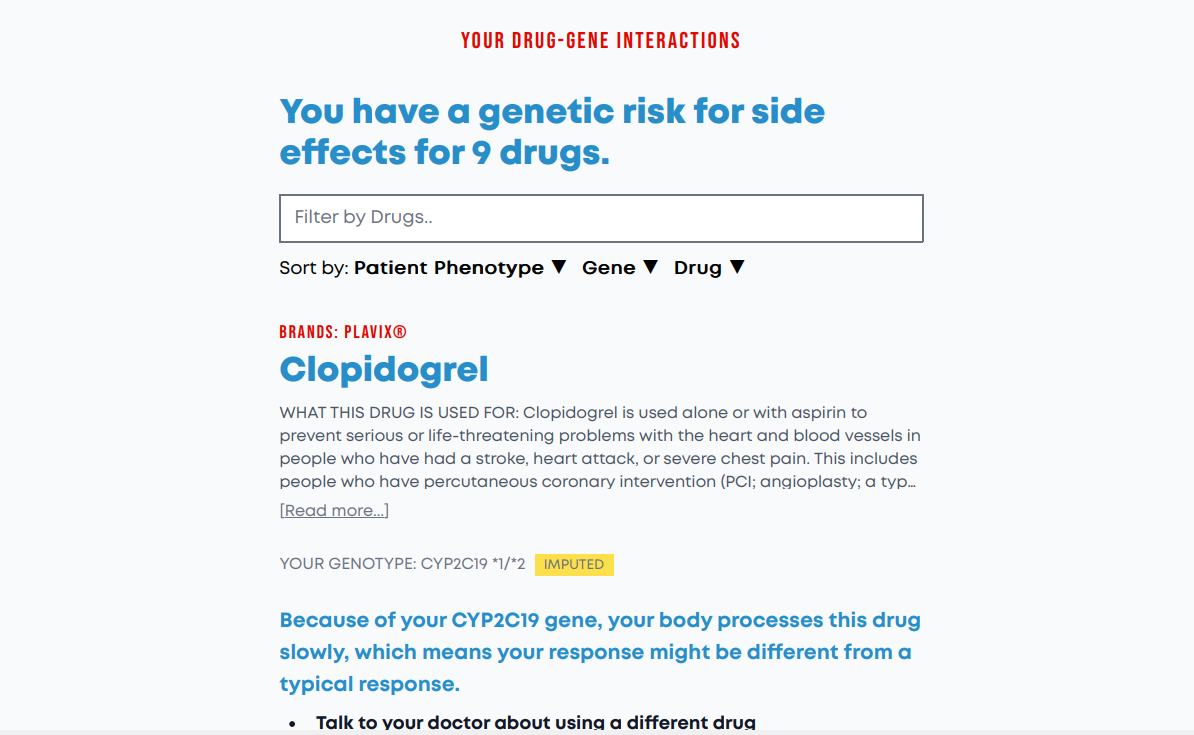
The FDA continually reviews pharmacogenomics research about how certain genetic markers and drugs work together to produce good responses from some people but not others. This information is already part of drug labeling for hundreds of drugs. Traitwell PGX takes that FDA-curated information about known drug-gene interactions, checks your DNA to see if you have those pharmacogenomically-relevant genetic markers, and gives you a list of drug-gene interactions customized for you. Rather than focusing too much on the technical details (e.g., star alleles) and medical jargon (e.g., “rapid metabolizer), Traitwell PGX helps you to identify specific drugs (including brand names) and learn what the FDA says about your risk of side effects given your genetic profile. You can also search your Traitwell PGX results by drug and sort them by response, gene, and drug. Each result has easy-to-understand actionable information (e.g., talk to your doctor about adjusting your dosage) to help you use your results.
Traitwell PGX is free if you’ve already tested your DNA with 23andMe, Ancestry DNA, My Heritage, Living DNA, or any of the various direct-to-consumer genotyping services. To use our app, download your raw DNA file from the service you already paid for, and upload it to Traitwell PGX to get your free results in minutes.
The usual caveats apply: Direct-to-consumer genetic tests are not clinical grade so that’s why “actionable” means you should use what you learn as a conversation starter with your doctor. Your doctor can then order confirmatory testing, which is important because direct-to-consumer genetic tests have high false-positive rates for rare genetic variants. Traitwell PGX is first and foremost an educational resource, not a diagnostic tool. Your results can also help you understand your personal experience with different drugs—for example, maybe codeine has always been too strong for you or maybe Dramamine never seems to work. Furthermore, your response to a drug depends not only on your genetic markers but also on your age, weight, liver and kidney function, your other medications, and your lifestyle.
Back in 2013, the FDA sent 23andMe a warning letter regarding its health reports. The FDA said that the company lacked the proper approval to give people potentially life-altering information about their health and that it needed to provide evidence that the results are reliable and won’t jeopardize consumers’ health. But 23andMe said that the health information in its reports was not meant to be diagnostic, just informative and educational. Nevertheless, the FDA disagreed. As a result, the company rescinded its health-related genetic testing services. Two years later, 23andMe began getting FDA approvals for genetic tests for breast cancer, Alzheimer’s disease, Parkinson’s disease, and other conditions. In 2018, 23andMe became the first company to get FDA approval for its pharmacogenomics reports. But perhaps wary of FDA scrutiny, 23andMe has scaled back these and other health reports.
The key issue is whether direct-to-consumer genotyping can provide accurate results for medical decisions.
At first glance, the situation looks unpromising. If genotyping produces poor results, doesn’t that mean drug-gene information from Traitwell PGX—like other third-party DNA analysis providers—is worthless? Studies show that genotyping false positive rates can range from 40% to as high as 85%. A false-positive is a test result that shows you have a disease or condition when you don’t actually have it, leading to unnecessary worry or counterproductive changes in medical treatment.
Even where the results are accurate, experts are still wary about direct-to-consumer pharmacogenomic reports. According to a 2017 analysis of direct-to-consumer pharmacogenomic testing, “One of the major concerns is the potential for misunderstanding and misinterpretation of the test results, particularly when pre- or post-test genetic counselling is not provided.” The real fear could be that you might modify your medications on your own.
In 2021, the FDA approved 23andMe’s pharmacogenomics report for two medications: clopidogrel, which is prescribed for certain heart conditions, and citalopram, which is prescribed for depression. To meet FDA requirements, 23andMe conducted studies designed to increase the likelihood of detecting the genetic variants associated with interactions with these two drugs and to reduce false-positive and false-negative results. These studies found that the accuracy of its pharmacogenetics reports exceed 99% concordance with Sanger sequencing, which is used in clinical genetic testing. So in order to get FDA approval, 23andme demonstrated that its genotyping results match up with clinical genetic testing. And it turns out that clinical genetic testing is merely a different type of genotyping.
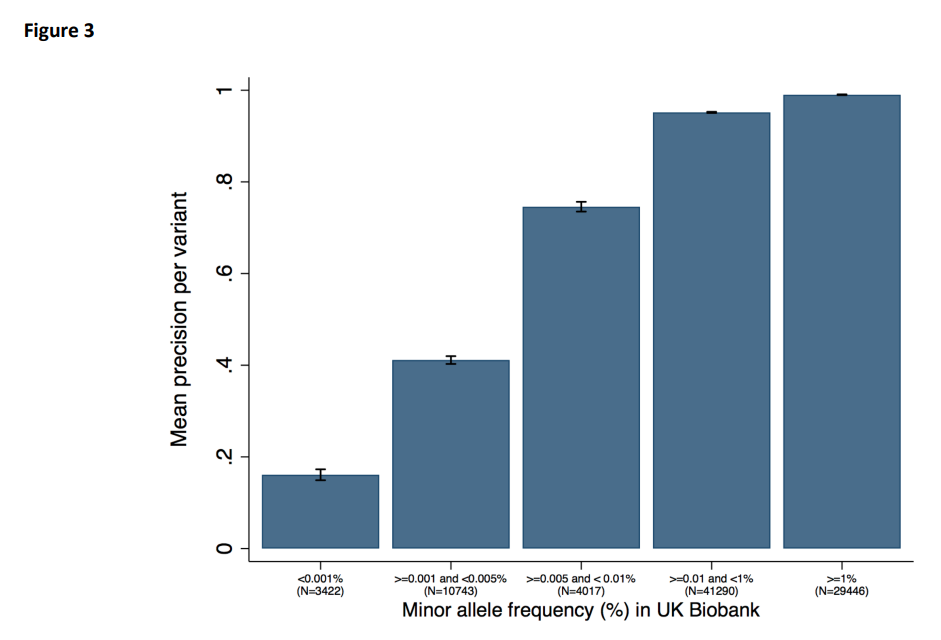
Genotyping services like 23andMe test only a subset of your genome. For looking at many different genetic variants at the same time, genotyping chips are an efficient, accurate, and cost-effective method. They do, however, require prior identification of the genetic variants of interest. For direct-to-consumer genetic testing services, it makes sense to focus on the variants that most people have. That’s why these services don’t do a good job of picking up rare variants—they’re just not designed that way.
In fact, genotyping chips are really quite good at picking up common genetic variants, i.e., variants found in at least 1 out of 100 individuals. A 2019 study found that the rates of true positives and true negatives are all above 99% for 108,574 common genetic variants. But the rarer the variant, the poorer the performance of genotyping chips. That’s why 23andMe got into trouble with the FDA for genetic tests for breast cancer—the BRCA1/2 genes involved are very rare.
If you are an average healthy person, that’s great news. It means that your direct-to-consumer genetic test is an excellent start. It means that Traitwell PGX can offer you decent preliminary results that can lead to fruitful discussions with your doctor or a useful educational resource that can validate your personal drug history. But you still need confirmatory testing through your doctor if you want to use drug-gene information for medical decision-making. That’s why we limit Traitwell PGX’s recommendations to statements like, “Talk to your doctor about adjusting your dosage.”
But the future actually lies in whole genome sequencing. Clinical-grade confirmatory genetic testing is still limited to analyzing a subset of your genome. It just targets a different subset of your genes. In 2021, a study found that whole-genome sequencing has low error rates related to pharmacogenomic testing. That’s because whole-genome sequencing is an analysis of your entire genome, not just a targeted subset. The accuracy is a matter of which technology is used to carry out the sequencing, and that continues to get better and less expensive over time. Furthermore, unlike confirmatory genetic testing, whole genome sequencing needs to be done only once, and then you are able to refer to it as your health needs change and as pharmacogenomic research advances. Think of it as your own lifelong health resource.
Despite causing an estimated 125,000 avoidable deaths each year and $100 billion annually in preventable health care costs, medication nonadherence is barely on the radar of most practicing physicians. Adherence rates for most medications for chronic conditions such as diabetes and hypertension usually fall in the 50% to 60% range, even with patients who have good insurance and drug benefits.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6045499/
An oft-cited reason that people don’t take their meds is side effects. No one enjoys taking medication and no one enjoys side effects. While doctors and other healthcare professionals are properly concerned with serious side effects like death and strokes, more mundane side effects like weight gain and nausea are much more relevant to your average patient. Quality of life is always a greater concern for yourself than for your doctor—after all, your doctor only needs to fix the immediate problem, but you must live with yourself during and after the process. One popular solution to this problem is, all too often, an app: offer an app to remind people to take their meds. Or give them fancy pill boxes with a nagging app built right in. However, efficacy rates are low for these approaches, despite the millions of dollars spent on their development.
So why not take side effects more seriously? If 40-50% of people are not taking their meds and if a big portion of that group doesn’t do it because of side effects, then maybe starting them off with medications or dosages with less risk of side effects can bump adherence rates up. Traitwell PGX is a drug-gene interaction reference tool that takes your raw DNA file and shows you the interactions that are relevant to your DNA. We’re putting pharmacogenomics information already available from the FDA, PharmGKB, CPIC, and other authorities into the hands of consumers and healthcare providers like you. For free. So try it out and let us know what you think.




It's a shame that I can't get in contact with anyone from traitwell to help me get my consanguinity score, been asking since July